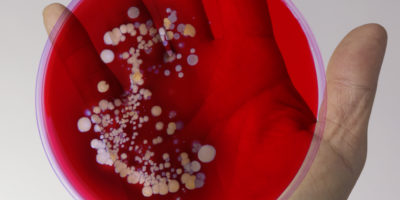
British Biotech Receives €52M Boost to Beat Antimicrobial Resistance with New Antibiotic
In a recent press release Summit Therapeutics announced they received the BARDA Contract worth $62 mil. for the development of a new treatment against Clostridium difficile infections.
Summit Therapeutics is a biopharma company operating in the US and UK with discovery, development and commercialization of novel medicines for diseases at its center.
Biomedical Advanced Research and Development Authority (BARDA) is part of US government’s Department of Health and Human Services’ Office of the Assistant Secretary for Preparedness and Response. The awarded contract will support the clinical development of ridinilazole, a new treatment for C. difficile infections. Ridinilazole is a new class of antibiotic with a narrow spectrum of activity, as it targets the harmful bacteria without damaging other useful bacteria in the human gut.
In a Phase 2 trial it was observed that ridinilazole is highly effective against C. difficile infection, while preserving the microbiome and substantially reducing disease recurrence.
Summit Therapeutics will first receive $32 mil. to commence the enrollment and dosing, and receiving the rest of the $30 mil. after achieving agreed performance-related targets.
Source: Summit Therapeutics
Read the full press release here.
Related updates

Important Pieces Of A Puzzle

Reflections On COMBACTE-NET