Funding
Both the European Commission and EFPIA invest in the Innovative Medicines Initiative (IMI), which supervises COMBACTE, allowing it to remain independent of commercial interests.
Improving clinical evaluation of new antibiotics
Research aimed at developing new medicines is usually financed by a single individual pharmaceutical sponsor collaborating with a single Clinical Research Oragnization (CRO). For each study a new set of clinical study sites are selected, and the collaboration ends when the study ends. In fact, these studies tend to be severely delayed, with many clinical sites not enrolling the expected number of study patients, sometimes with compromised quality. The 4 COMBACTE projects are collaborations between pharmaceutical companies (EFPIA partners) and academic partners, so-called public-private partnerships) with the aim to improve the efficency of the clinical evaluaton of new antimicrobial agents.
IMI’s New Drugs for Bad Bugs (ND4BB) program
IMI developed the New Drugs for Bad Bugs program to combat multi-drug resistant bacteria. It focuses entirely on incentivizing the development of new antibacterial drugs. All the details, including more information about the Translocation, iABC, Enable and Drive A/B projects can be found on IMI.
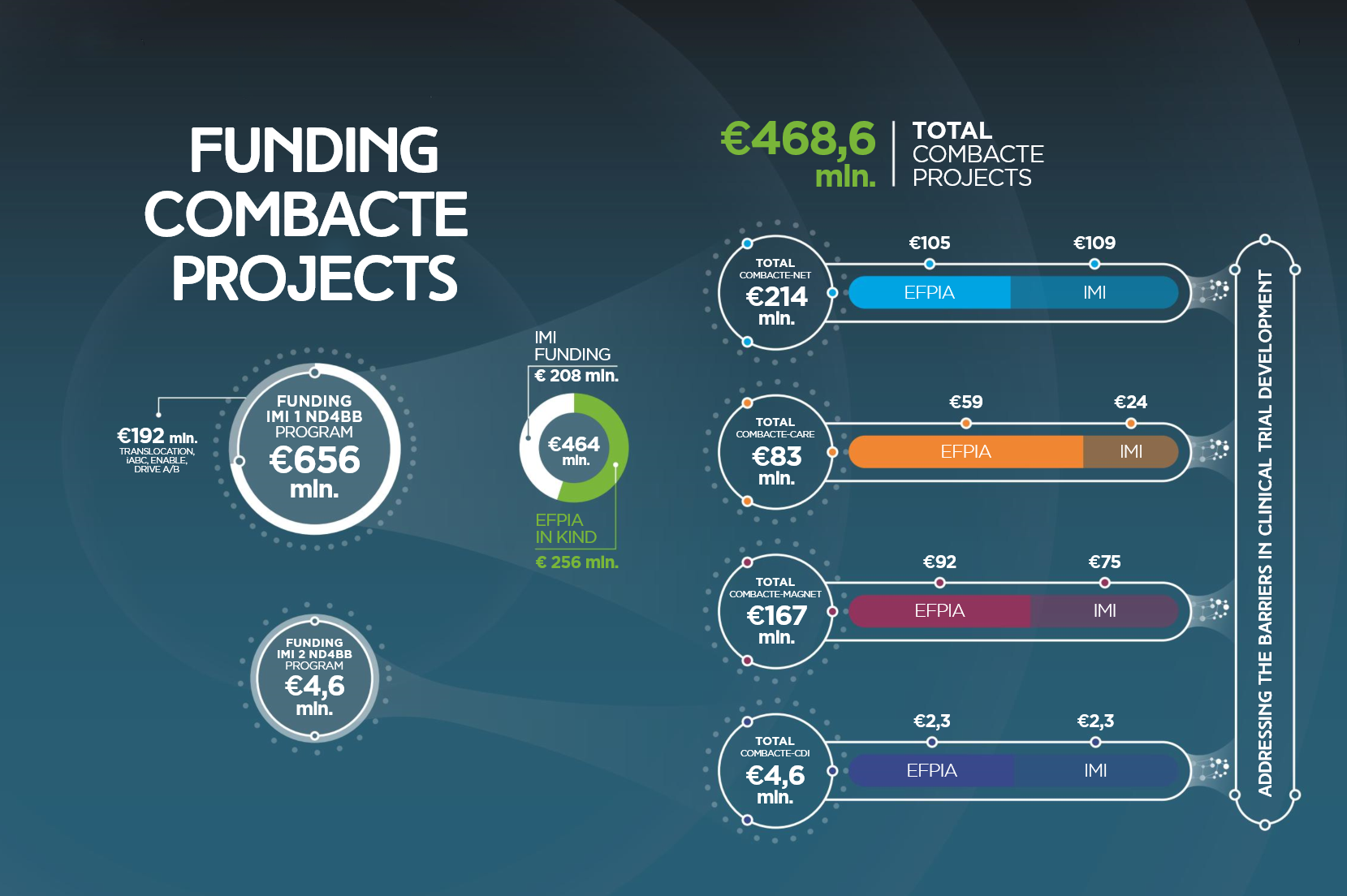
Source of COMBACTE’s budget
IMI 1 ND4BB
€208 million from the EU
€256 million from the EFPIA
€464 million in total
IMI 2 ND4BB
€2.3 million from the EU
€2.3 million from the EFPIA
€4.6 million in total
Utilization of COMBACTE’s budget
IMI 1 ND4BB Program
€214 million on COMBACTE-NET
€83 million on COMBACTE-CARE
€167 million on COMBACTE-MAGNET
€464 million in total
IMI 2 ND4BB Program
€4,6 million on COMBACTE-CDI