WP2 – Current Practices
Assess current practices in regards to CDI diagnosis and treatment in Europe and their potential impacts, including guidelines, testing, surveillance, treatments and costs.
Study objectives
Objective 2.1 – To highlight current guidelines for disease detection, notification and management/control
Objective 2.2 – To determine the heterogeneity in testing density and in following the existing guidance (including identification of missed diagnoses and empiric treatment approach practices)
Objective – 2.3 To determine the heterogeneity in current surveillance practices and ways to improve them
Objective – 2.4 To describe current treatment pathways for patients with CDI infection and recurrences
Objective – 2.5 To elucidate the overall costs of CDI, including management of multiples episodes and re-hospitalization (healthcare perspectives including indirect financial impact on the healthcare system)
Objective 2.6 – To measure the impact of CDI management on the healthcare system and the impact of current control measures (infection control/antibiotic stewardship)
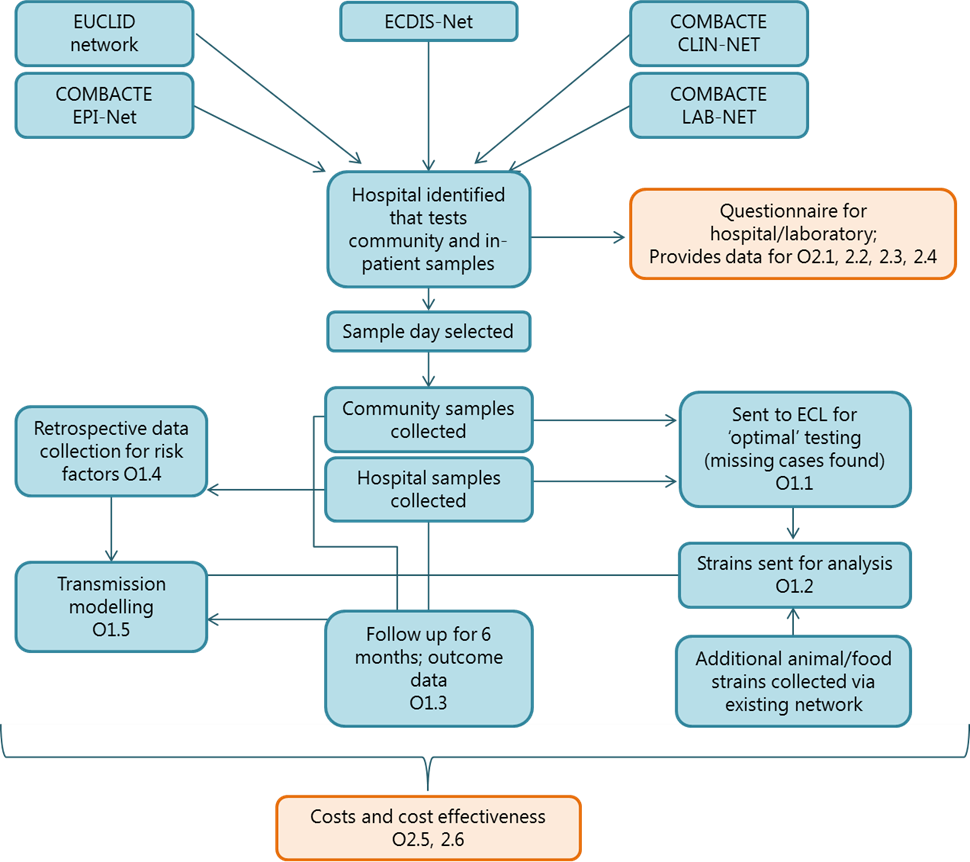
Schematic representation of study design (WP1 &WP2)
A synergistic approach will enable completion of all the objectives from WP1 and WP2, with data created from a single, large complex study flowing into other objectives for further results analysis. Boxes in blue denote objectives in WP1, and those in orange denote for objectives in WP2. Due to the inter-related nature of the objectives within both WP1 and WP2, the flow of work has been planned to ensure maximum outputs within the timeframe of the grant. The directions of the arrows indicates data flow and, therefore, also the timing of activities. This ambitious comprehensive project design takes influence from several existing, separate studies; the scope of producing all of these data from one, well-designed and far-reaching study is novel and innovative.
Related updates
Reflecting on COMBACTE-CDI
